About half the human brain mass is lipid. Several brain disorders are known to be caused by abnormal lipid metabolism, disruptions in the processes of making lipids and breaking them down. Second only to Alzheimer’s in prevalence is frontotemporal dementia (FTD), one form of which encompasses a range of social, behavioral, or language disorders (as opposed to memory or motor deficits seen in other disorders). Several genes have been associated with FTD, foremost among them the conserved genes MAPT (tau), PSEN1 (presenilin), VCP (valosin containing protein) and GRN (granulin).
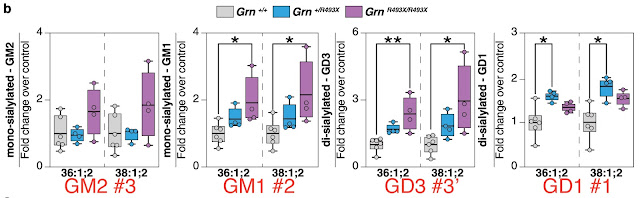
Granulins are a family of secreted, glycosylated peptides (A, B, C, etc.) cleaved from a single precursor (progranulin, PGRN), that are involved in a wide range of activities probably due to their roles in regulating protein lysosomal protein metabolism. The authors of this study found that gangliosides (i.e., glycosphingolipids with attached sialic acids) are elevated in brains of granulin mutant mice (GRN R504X), which are analogous to the most prevalent granulin mutation in humans, R493X (substitution of the arginine normally at position 493 with a nonsense codon, resulting in a truncated protein). This mutation causes neuronal ceroid lipofuscinosis, a severe neurodevelopmental disease, in humans and neuroinflammation in mice (Jax). The metabolic order in the ganglioside degradation pathway (Fig. 1a) is first disialylated GD1 (Fig. 1b, shown, rightmost plot, annotated with red #1) 👉 monosialylated GM1 (#2) 👉 GM2 (#3) 👉 GM3. Also, GD2 👉 GM3 (#3’) via an alternative pathway. Levels of precursor GD1 (#1) are significantly elevated in the brains of mice with heterozygous mutant granulin (Figure 1b: blue fill, Grn +/R493X) compared with normal granulin (Grn+/+, grey) but not in homozygous mutants (purple). This may suggest a feed-back mechanism that limits the accumulation of that metabolite. GM1, #2, is elevated in homozygous mutant brain compared to normal, with the heterozygous mutant intermediate. However, GM2, #3, is not significantly elevated in mutation-bearing mouse brains. The alternate pathway, #3’, shows elevated GD3, the precursor to GM3, in homozygous mutant brains.
They also analyzed the lipids in postmortem human brains of 12 GRN mutation-related FTD cases, 6 sporadic FTD cases, and 3 control normal subjects. GD3 and GM1 are significantly elevated in GRN-related FTD cases (Fig 1c, blue columns). However, they both seem also elevated in non-GRN (sporadic) FTD cases (green). GD1 is significantly elevated in GRN-related but not -unrelated FTD.
In a striking simplification, they tested the effects of removing the granulin precursor protein gene, PGRN (same as GRN), in HeLa cells (Fig 2). They found elevated GM2 in the deficient line (GRN-/-) that was reduced to normal levels by restoring granulin (GRN-/- + PGRN-addback).
What causes changes in the levels of gangliosides? Gangliosides are catabolized by lysosomal enzymes. However, those enzymes were not altered by GRN deficiency (Fig 3). An intermediate metabolite, bis(monoacylglycero)phosphate (BMP), which is crucial to ganglioside degradation, was found to be reduced by 50-60% in GRN-deficient HeLa cells and mouse brains, and ‘markedly’ in human brains of FTD cases, although again both GRN-related and sporadic cases (Fig 4d). The authors propose a model wherein “lysosomal granulin peptides maintain lysosomal function and homeostasis, including the levels of BMP, that are crucial for ganglioside catabolism”. Their results await confirmation by others and many details remain to be pursued further. One relatively simple aspect will be clarifying how the model accounts for autosomal dominance of GRN deficiency. Also worth noting is the proximity of GRN and the Alzheimer- and Parkinson-associated MAPT genes, within a million nucleotides in band 17q21.31.
Boland S, Swarup S, Ambaw YA, Malia PC, Richards RC, Fischer AW, Singh S, Aggarwal G, Spina S, Nana AL, Grinberg LT, Seeley WW, Surma MA, Klose C, Paulo JA, Nguyen AD, Harper JW, Walther TC, Farese RV Jr. Deficiency of the frontotemporal dementia gene GRN results in gangliosidosis. Nat Commun. 2022 Oct 7;13(1):5924. doi: 10.1038/s41467-022-33500-9. PMID: 36207292; PMCID: PMC9546883.




No comments:
Post a Comment