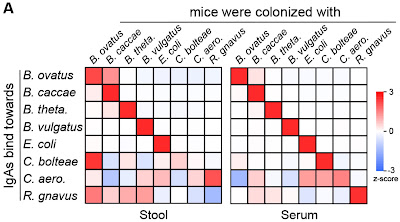
Not surprisingly, the antibodies tend to bind specifically to those strains of microbes against which they were stimulated (shown, Figure 1, panel A). They also found that mice monocolonized from birth produce more IgA reactive with that species (termed ‘self’) than newly-introduced species. They make a point about IgA being able to ‘aggregate pathogenic bacteria’ and ‘selectively coat disease-associated bacteria’ but it is unclear how IgA itself could distinguish dangerous from benign and anyway they tested only benign bacteria.
A small panel (29) of monoclonal IgA antibodies cloned from gut tissues of monocolonized mice also showed species specificity. Finally, they showed that monoclonal IgA antibodies with specific binding activity could be detected in the feces of mice that had been force-fed the IgA 3 hours previously, suggesting a targeted, potential therapy (e.g., against the human pathogen Clostridioides difficile).