Fumarate hydratase (FH), also known as fumarase, catalyses hydration of fumarate to malate and is found in mitochondria, where it performs in the Krebs cycle, and the cytosol, where it performs in the urea cycle and in catabolizing amino acids. It is encoded by homologous genes in bacteria, yeast, and eukaryotes. Fumarase deficiency in humans is involved in a range of symptoms from neurological abnormalities in newborns to tumors in adults.
These authors examined what happens immediately after loss of FH. They generated a mouse line with floxed Fh1 gene (homolog of the human FH) and crossed it with a line with ubiquitous, inducible, Cre recombinase (Rosa26). Induction of Cre caused the genetic loss of FH expression within 5 days and metabolic changes within 10 days (Fig 1). They found mitochondria swell and lose their genome (mtDNA) into the cytosol, where it triggers innate immunity (STING) and stimulates inflammation (RIG-I) including expression of interferon stimulated genes (ISGs).
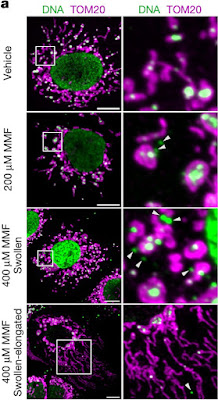 Treating cultured cells with a cell-permeable derivative of fumarate, monomethylfumarate (MMF), caused similar releases of mtDNA into the cytosol and expression of ISGs (Fig 3 Shown), strongly suggesting that the accumulation of fumarate causes the changes observed after loss of FH. They also show that mtDNA is released through ‘mitochondria-derived vesicles’ (MDVs), normally used to transport ‘content without affecting the integrity of the membranes’ (Fig 4), so the process occurs under some control. Finally, they show activation of DNA-sensor and innate immunity pathways in FH-deficient renal cancers, suggesting relevance for humans.
Treating cultured cells with a cell-permeable derivative of fumarate, monomethylfumarate (MMF), caused similar releases of mtDNA into the cytosol and expression of ISGs (Fig 3 Shown), strongly suggesting that the accumulation of fumarate causes the changes observed after loss of FH. They also show that mtDNA is released through ‘mitochondria-derived vesicles’ (MDVs), normally used to transport ‘content without affecting the integrity of the membranes’ (Fig 4), so the process occurs under some control. Finally, they show activation of DNA-sensor and innate immunity pathways in FH-deficient renal cancers, suggesting relevance for humans.Fig 3a, Fumarate causes mitochondrial changes. Overlay panels. Cells were treated for 8 days with solvent alone (vehicle) or with fumarate (200 or 400 uM MMF doses) then stained for DNA (green) and mitochondria (outer membrane protein TOM20, purple). Scale bars 10 um. Boxed area in left column expanded in right column.
One wonders what happens after the loss of other ancient and obscurely important cellular infrastructure.
Zecchini V, Paupe V, Herranz-Montoya I, Janssen J, Wortel IMN, Morris JL, Ferguson A, Chowdury SR, Segarra-Mondejar M, Costa ASH, Pereira GC, Tronci L, Young T, Nikitopoulou E, Yang M, Bihary D, Caicci F, Nagashima S, Speed A, Bokea K, Baig Z, Samarajiwa S, Tran M, Mitchell T, Johnson M, Prudent J, Frezza C. Fumarate induces vesicular release of mtDNA to drive innate immunity. Nature. 2023 Mar;615(7952):499-506. doi: 10.1038/s41586-023-05770-w. Epub 2023 Mar 8. PMID: 36890229; PMCID: PMC10017517.